Am I using the wrong equation? This question threw me off as it isn't circular motion like many magnetic field problems. I thought I was on the right track by dividing (2 $\times$ the charge of an electron $\times$ the voltage) by the mass of an electron, and then square rooting everything. On the submicroscopic scale, it is more convenient to define an energy unit called the electron volt (eV), which is the energy given to a fundamental charge accelerated through a potential difference of 1 V. $F=Bqv$, where F is the force of the magnetic field, B is the magnetic field strength, q is the charge and v is the velocity. The sum of the charges of quarks that make up a nuclear particle determines its electrical charge. This fundamental property of matter is known as electric charge. The following equations are the ones we've been using in class to solve magnetic field problems: This means that electrons have a negative charge (-1) while protons have a positive charge ( 1). The wires produce a magnetic field of up to $0.68 T$. 
Coulomb's law works well for predicting the energy of an electron in a hydrogen atom (H has only two particles: one nucleus and one electron). The electrons are accelerated to a voltage of $3 kV$ by wire coils and are then directed to different points on the screen. According to Coulomb's law, the attraction of an electron to a nucleus depends only on three factors: the charge of the nucleus ( Z), the charge of the electron (-1), and the distance between the two (r). In the SI system of units, the value of the elementary charge is exactly defined as e īy combining the best measured value of the antiproton charge (below) with the low limit placed on antihydrogen's net charge by the ALPHA Collaboration at CERN.So I'm trying to solve this problem in which an electron beam is "drawing a picture" on a TV screen. This elementary charge is a fundamental physical constant. 
But if you observe the structure of an atom, neutrons(neutral), and. The elementary charge, usually denoted by e, is the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric charge carried by a single electron, which has charge −1 e. According to science, like charges repel each other and opposite charges attract each other. 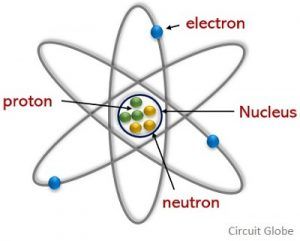
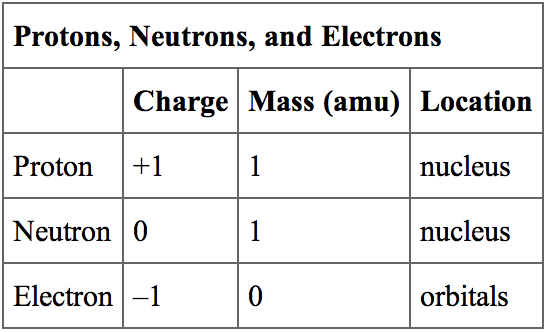
Charge carried by one proton or electron Elementary charge 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
